Nag sag pms6/24/2023 
we explain how to tell if a salt is soluble or insoluble. often students are required in this video we go over solubility rules for salts. but, when two ions in two different the solubility rules are important for quickly figuring out is a substance is soluble or insoluble in water. this video shows how ionic compounds dissolve in water, dissociating into individual ions. both are based on solubility rules in the form of a song. it contains a table or chart of the the solubility chart and solubility rules are two ways to determine if a substance is soluble in water. this chemistry video tutorial focuses the difference between soluble and insoluble compounds. it really goes a long way! □ subscribe: learn how to memorize solubility rules very easily. please subscribe and hit that thumbs up button. (it shall help you with your so solubility can be difficult if you don't know how to properly use a solubility table! in this video i go over all of the solubility rules this chemistry video tutorial explains how to use the solubility rules to determine if a compound is soluble or insoluble. compounds with group 1 cations and nh 4.Īnnoyed by all those kids sagging their pants? well, here's a good way to put that disturbance to use.even so, you will save a lot of time (now and later in ut chem classes) if you commit these two solubility rules to memory: feel free to have this table with you when you take the assessment or when you are in learning mode in aleks. This is the solubility table that is available for use within aleks. while memory questions are typically found as free standing questions, they can also be tucked into a passage and represent 25% of science questions on the mcat. 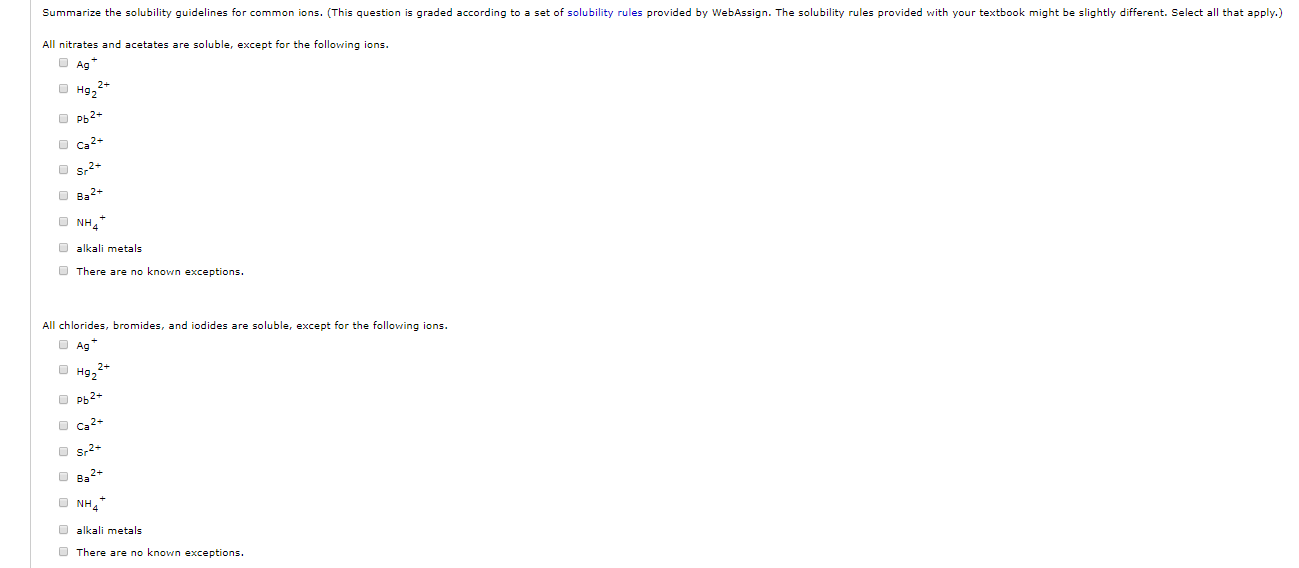
memory questions on the mcat are exactly what they sound like: they test your knowledge about a specific fact or concept. alkali (group 1a) salts and nh 4 are soluble. there is no short cut! all soluble except with c hlorates acetates sulfates c ab as r h ga gp b (cbs happy) h alogens h ga gp b (happy) n’ itrates g roup (ia) i a simple solubility rules: nitrate (no 3 ) salts are soluble. 
Way to remember these is pure memorization. agno3 and ag (c2h3o2) are common soluble salts of silver virtually all others are insoluble. thus, agcl, pbbr2, and hg2cl2 are insoluble. important exceptions to this rule are halide salts of ag, pb2, and (hg2)2. Salts containing cl, br, or i are generally soluble. Solubility Rules Chart For Chemistry Classroom 11th Chemistry
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
